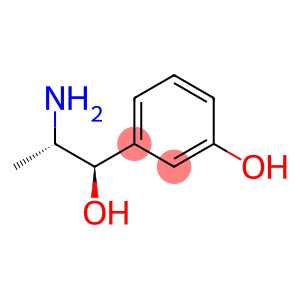
alpha-(1-Aminoethyl)-m-hydroxybenzyl alcohol
metaraminol
CAS: 54-49-9
Molecular Formula: C9H13NO2
alpha-(1-Aminoethyl)-m-hydroxybenzyl alcohol - Names and Identifiers
alpha-(1-Aminoethyl)-m-hydroxybenzyl alcohol - Physico-chemical Properties
| Molecular Formula | C9H13NO2 |
| Molar Mass | 167.21 |
| Density | 1.1222 (rough estimate) |
| Melting Point | 107.5°C |
| Boling Point | 295.79°C (rough estimate) |
| pKa | pKa 8.6 (Uncertain) |
| Refractive Index | 1.4760 (estimate) |
| Physical and Chemical Properties | The tartaric acid salt of the product is a white crystalline powder. Melting point 176-177 °c. Easily soluble in water, the pH of 1% aqueous solution is about 3.5. Slightly soluble in ethanol, insoluble in chloroform, ether. Almost odorless and bitter. |
| Use | 2-adrenergic receptor agonist, suitable for all kinds of Shock and low blood pressure during surgery |
alpha-(1-Aminoethyl)-m-hydroxybenzyl alcohol - Reference Information
| Overview | M-hydroxylamine mainly acts on α receptor, but weakly on β1 receptor; part of the effect is by promoting the release of norepinephrine from sympathetic nerve endings. It is suitable for the treatment of early Shock, and the prevention and treatment of acute hypotension during spinal anesthesia. It can also be used for adjuvant symptomatic treatment of hypotension due to bleeding, drug allergy, surgical complications and brain trauma or brain tumor with Shock, and also for the treatment of hypotension due to cardiogenic Shock or sepsis. |
| Use | is an adrenergic mimetic drug, used for acute hypotension, and is one of the commonly used drugs for rescuing patients. |
| production method | M-hydroxybenzaldehyde (see 21680) is prepared by enzymatic addition, condensation and catalytic hydrogenation to obtain m-hydroxylamine. The M-hydroxylamine ethanol solution was added with tartaric acid to form a salt, and the precipitated crude product was recrystallized with anhydrous and no alcohol to obtain m-hydroxylamine bitartrate. |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: METARAMINOL Request for quotation
CAS: 54-49-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 54-49-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: METARAMINOL Request for quotation
CAS: 54-49-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 54-49-9
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
View History
